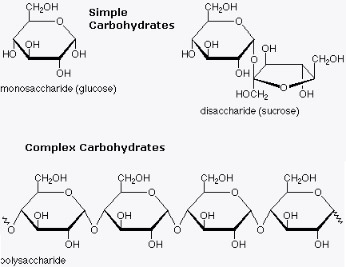
Carbohydrate
Carbohydrates are technically hydrates of carbon; structurally it is more accurate to view them as polyhydroxy, aldehydes and ketones. The term is most common in biochemistry, where it is a synonym of saccharide, a group that includes sugars, starch, and cellulose. All carbohydrates can be classified as either monosaccharides, oligosaccharides or polysaccharides. Anywhere from two to ten monosaccharide units, linked by glycosidic bonds, make up an oligosaccharide. Polysaccharides are much larger, containing hundreds of monosaccharide units. The presence of the hydroxyl groups allows carbohydrates to interact with the aqueous environment and to participate in hydrogen bonding, both within and between chains. Derivatives of the carbohydrates can contain nitrogens, phosphates and sulfur compounds. Carbohydrates also can combine with lipid to form glycolipids or with protein to form glycoproteins
Carbohydrates have six major functions within the body:
- Providing energy and regulation of blood glucose
- Sparing the use of proteins for energy
- Breakdown of fatty acids and preventing ketosis
- Biological recognition processes
- Flavor and Sweeteners
- Dietary fiber
MONOSACCHARIDES
Monosaccharides are the simplest units of carbohydrates and the simplest form of sugar. They are the building blocks of more complex carbohydrates such as disaccharides and polysaccharides. Physically, they are usually colorless, can dissolve in water, and have the appearance of a crystal-like substance.
Functional GroupsMonosaccharides are classified as well based on their functional groups. A functional group is categorized by atoms or bonds that are responsible for the chemical reactivity within a molecule.
If a monosaccharide contains a ketone group in an inner atom, then the monosaccharide is classified as a ketose. A ketone group is a carbon atom forming a double bond with oxygen and single bonds with two hydrocarbon groups. A hydrocarbon group is a group that contains carbon bonded with hydrogen.
If a monosaccharide contains an aldehyde group at an end carbon, meaning a carbon at the end of the chain in the Fischer projection, then the monosaccharide is classified as an aldose. An aldehyde group is a carbon atom forming a double bond with oxygen and a single bond with hydrogen.
Monosaccharides are the simplest units of carbohydrates and the simplest form of sugar. They are the building blocks of more complex carbohydrates such as disaccharides and polysaccharides. Physically, they are usually colorless, can dissolve in water, and have the appearance of a crystal-like substance.
Functional GroupsMonosaccharides are classified as well based on their functional groups. A functional group is categorized by atoms or bonds that are responsible for the chemical reactivity within a molecule.
If a monosaccharide contains a ketone group in an inner atom, then the monosaccharide is classified as a ketose. A ketone group is a carbon atom forming a double bond with oxygen and single bonds with two hydrocarbon groups. A hydrocarbon group is a group that contains carbon bonded with hydrogen.
If a monosaccharide contains an aldehyde group at an end carbon, meaning a carbon at the end of the chain in the Fischer projection, then the monosaccharide is classified as an aldose. An aldehyde group is a carbon atom forming a double bond with oxygen and a single bond with hydrogen.
DISACCHARIDES
Disaccharide is two monosaccharides combined into one. More specifically, a disaccharide results when two monosaccharides are joined in a chemical process called dehydration synthesis (i.e., two monosaccharides combine, losing a water molecule in the process). This process is also known as a condensation reaction.
All carbohydrates are made up of carbon, hydrogen and oxygen and follow the chemical formula Cx(H20)y (e.g., C6H12O6 or C12H22O11).
Common Disaccharides
Disaccharides are generally water-soluble and sweet when ingested. The most commonly encountered disaccharides are listed below, along with the two monosaccharides from which they are made. Disaccharides are also known as complex sugars. Again, sugars are created through dehydration synthesis (aka condensation reaction) and broken down through hydrolysis, a process usually aided by enzymes.
Disaccharide is two monosaccharides combined into one. More specifically, a disaccharide results when two monosaccharides are joined in a chemical process called dehydration synthesis (i.e., two monosaccharides combine, losing a water molecule in the process). This process is also known as a condensation reaction.
All carbohydrates are made up of carbon, hydrogen and oxygen and follow the chemical formula Cx(H20)y (e.g., C6H12O6 or C12H22O11).
Common Disaccharides
Disaccharides are generally water-soluble and sweet when ingested. The most commonly encountered disaccharides are listed below, along with the two monosaccharides from which they are made. Disaccharides are also known as complex sugars. Again, sugars are created through dehydration synthesis (aka condensation reaction) and broken down through hydrolysis, a process usually aided by enzymes.
- Sucrose (table sugar) comes from glucose and fructose joining
- Lactose (milk sugar) comes from glucose and galactose joining
- Maltose (malt or beer sugar) comes from two glucose molecules joining
POLYSACCAHARIDES
Polysaccharides [Greek poly = many; sacchar = sugar] are complex carbohydrates, composed of 10 to up to several thousand monosaccharides arranged in chains. The most common monosaccharide in polysaccharides are glucose, fructose and galactose.
3 main polysaccharides related to the human nutrition include:
Non-digestible polysaccharides or dietary fiber, such as cellulose, promote the passage of food through the gut and thus help maintain bowel regularity. Some non-digestible polysaccharides, such as inulin, may also promote the growth of beneficial intestinal bacteria.
None of the polysaccharides are essential nutrients; you do not need to consume them in order to be healthy.
Polysaccharides [Greek poly = many; sacchar = sugar] are complex carbohydrates, composed of 10 to up to several thousand monosaccharides arranged in chains. The most common monosaccharide in polysaccharides are glucose, fructose and galactose.
3 main polysaccharides related to the human nutrition include:
- Starch ─ an energy source obtained from plants
- Cellulose ─ a structural polysaccharide in plants; when consumed, it acts as a dietary fiber
- Glycogen ─ a storage form of glucose in the human liver
Non-digestible polysaccharides or dietary fiber, such as cellulose, promote the passage of food through the gut and thus help maintain bowel regularity. Some non-digestible polysaccharides, such as inulin, may also promote the growth of beneficial intestinal bacteria.
None of the polysaccharides are essential nutrients; you do not need to consume them in order to be healthy.
CARBOHYDRATE METABOLISM
The entire spectrum of chemical reactions, occur in living systems are referred as metabolism. Types of metabolic pathways:
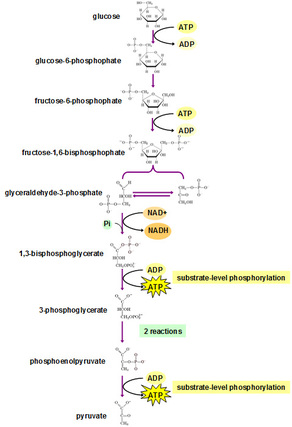
1) Glycolysis
Sequence of reactions of glucose to lactate & pyruvate with the production of ATP.
Site: Cytosolic fraction of cell
2) Glyconeogenesis
Generation of glucose from non-carbohydrate carbon substrates such as pyruvate, lactate, glycerol, and glucogenic amino acids.
Site: Liver and Kidney
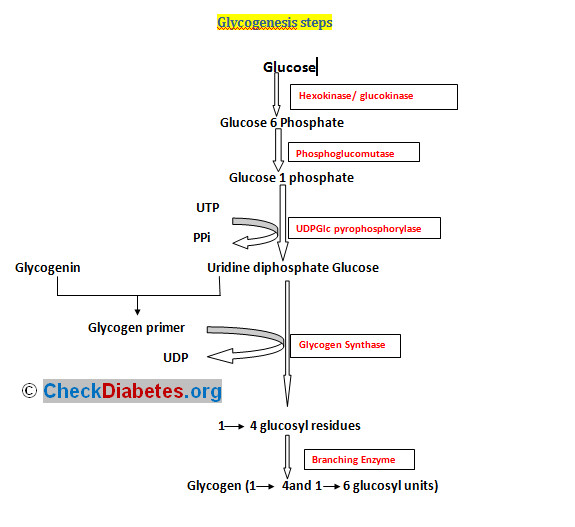
3) Glycogenesis
Biosynthesis of glycogen, major storage of carbohydrate.
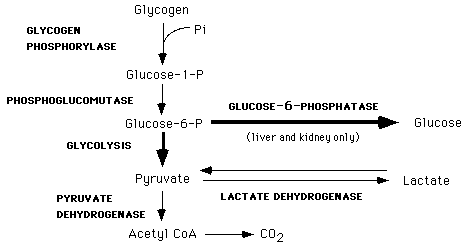
4) Glycogenolysis
Breakdown of glycogen (n) to glucose-6-phosphate and glycogen (n-1). Glycogen branches are catabolized by the sequential removal of glucose monomers via phosphorolysis, by the enzyme glycogen phosphorylase.
5) Pentose Phosphate Pathway
Metabolic pathway parallel to glycolysis that generates NADPH and pentoses (5-carbon sugars)
- anabolic pathways: protein synthesis
- catabolic pathways: oxidative phosphorylation
- amphibolic pathways: citric acid cycle
1) Glycolysis
Sequence of reactions of glucose to lactate & pyruvate with the production of ATP.
Site: Cytosolic fraction of cell
2) Glyconeogenesis
Generation of glucose from non-carbohydrate carbon substrates such as pyruvate, lactate, glycerol, and glucogenic amino acids.
Site: Liver and Kidney
3) Glycogenesis
Biosynthesis of glycogen, major storage of carbohydrate.
4) Glycogenolysis
Breakdown of glycogen (n) to glucose-6-phosphate and glycogen (n-1). Glycogen branches are catabolized by the sequential removal of glucose monomers via phosphorolysis, by the enzyme glycogen phosphorylase.
5) Pentose Phosphate Pathway
Metabolic pathway parallel to glycolysis that generates NADPH and pentoses (5-carbon sugars)